The Future of Prosthetics and Brain-Machine Interfaces
A groundbreaking neural implant, developed by bio-engineers from the University of Cambridge, has the potential to restore limb function in paralyzed limbs.
The revolutionary device combines flexible electronics tech and human stem cells, overcoming the limitations of previous neural implants, and offering new possibilities for prosthetics, brain-machine interfaces, and enhanced cognitive abilities.
Up to now, traditional neural implants have faced obstacles, primarily due to scar tissue formation that disrupts the connection between the device and the nerve.
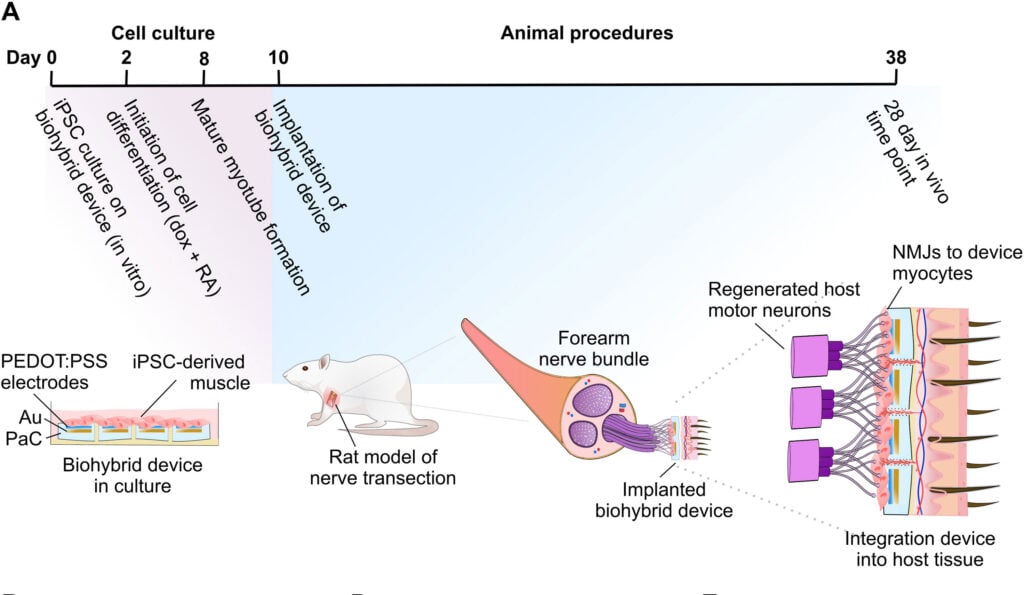
The Cambridge team’s innovative biohybrid device addresses this issue by incorporating human stem cells that are reprogrammed into muscle cells, which then create a layer between the electrodes and living tissue. This prevents scar tissue formation and allows for seamless integration with the host’s body.
The biohybrid device consists of a biocompatible flexible electronic device which is just about thin enough to be attached to the end of a nerve. This uses a layer of induced pluripotent stem cells (iPSC), which have been reprogrammed into muscle cells and then placed on the electrode. This is the first time iPSCs have been used in such a manner in a living organism, offering an unprecedented level of control and adaptability.
Revolutionizing Prosthetics and Brain-Machine Interfaces
The team tested their new technology by implanting the biohybrid device into the paralyzed forearm of rats, where it was able to pick up signals from the brain controlling movement. Although the rats’ forearms remained immobile, the device demonstrated the potential to restore function once connected to the rest of the nerve or a prosthetic limb.
Fabrication and implantation process of the biohybrid device: Stem cells are seeded onto the device, transformed into muscle cells, and implanted into a rat nerve model for 4 weeks.
There are a few obvious advantages of this new neural implant. Besides its easy integration and long-term stability, the device’s small size means that implantation requires only minimally invasive keyhole surgery. It could also be used to control prosthetic limbs or improve brain-machine interfaces, truly revolutionizing the way we interact with technology.
The Cambridge team is now working on optimizing the devices and has filed a patent application for the technology.
The study, published in the journal Science Advances, demonstrates the first step toward restorative therapies using regenerative bioelectronics. By enhancing the tissue-electronics interface with an intermediate cell layer, the researchers have achieved improved resolution and electrical recording in vivo.
The development of this biohybrid neural implant is a major leap forward in prosthetics and brain-machine interfaces. By combining living human cells with bioelectronic materials, the device can communicate with the brain more naturally and intuitively, offering new possibilities for enhancing neurological functions in the nervous system.
TLDR
- Researchers from the University of Cambridge have developed a groundbreaking neural implant that could restore limb function in paralyzed limbs.
- The biohybrid device combines flexible electronics and human stem cells to overcome limitations of previous neural implants and improve prosthetics, brain-machine interfaces, and cognitive abilities.
- Induced pluripotent stem cells (iPSC) are used to prevent scar tissue formation and allow seamless integration with the host’s body.
- The device has been successfully tested in paralyzed rat forearms, demonstrating its potential to restore function once connected to a nerve or prosthetic limb.
- Advantages include easy integration, long-term stability, and minimally invasive keyhole surgery for implantation.
- Researchers are working on optimizing the device and have filed a patent application for the technology.
- This development is a major leap forward in prosthetics and brain-machine interfaces, offering new possibilities for enhancing neurological functions in the nervous system.








